In a groundbreaking advancement for environmental chemistry, a dedicated research team from UNIST has unveiled a catalyst that not only replicates the functionality of natural enzymes but also offers a promising solution for tackling hazardous hydrocarbons. Led by Professor Jaeheung Cho, this team has successfully engineered a catalyst that emulates metalloenzymes, which play a pivotal role in various biological processes. Their work, recently published in the Journal of the American Chemical Society, marks a considerable step toward greener chemistry practices by enhancing our ability to oxidize harmful pollutants in an energy-efficient manner.
Innovative Mechanisms at Play
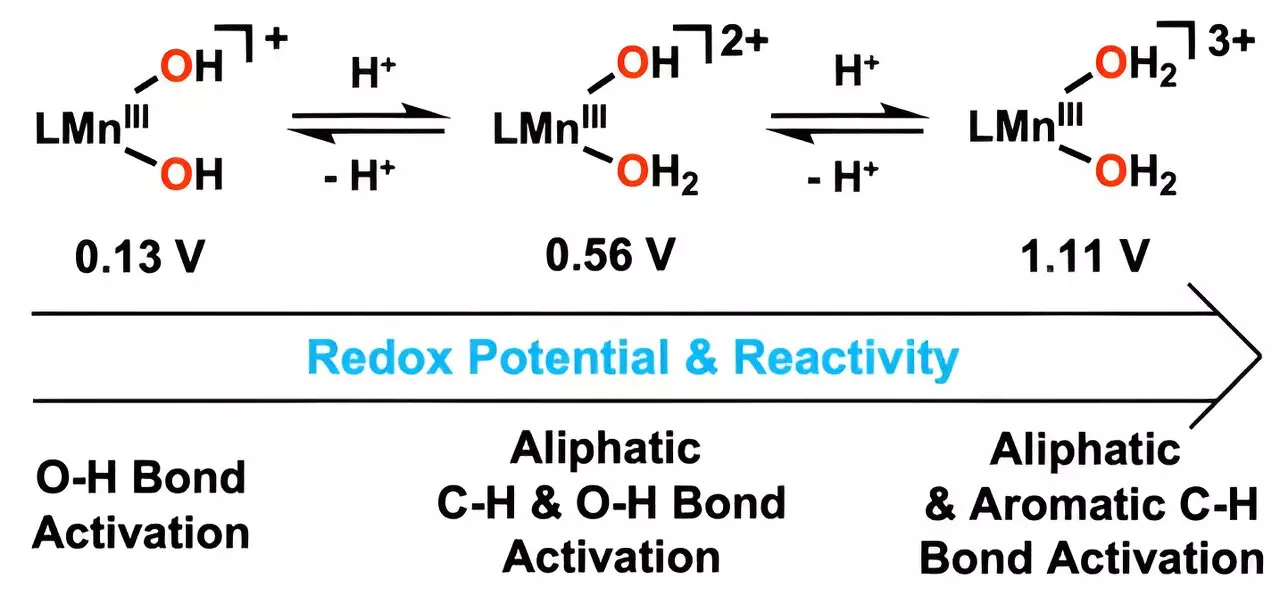
The team’s innovative approach involved adding hydrogen ions to hydroxo ligands, resulting in the synthesis of metal-bound water molecules. This novel configuration empowers the catalyst to oxidize carbon-hydrogen (C-H) bonds at significantly lower temperatures compared to traditional methods. The implications of this technological leap are vast; by operating at such reduced temperatures, energy consumption is minimized, leading to both economic and environmental benefits. Such advancements signal a shift toward sustainable practices that can be adopted on a larger scale, potentially revolutionizing industrial applications.
Enhancing Reaction Rates with Manganese
At the heart of this new catalyst’s effectiveness lies the transformation of manganese’s reduction potential. By intricately modifying the hydroxyl ligand to incorporate water, the catalyst demonstrates improved capabilities in activating oxygen-hydrogen bonds. Such alterations have been crucial in achieving accelerated reaction rates, leading to more efficient breakdown of complex hydrocarbons. The fact that this catalyst has shown remarkable efficiency in oxidizing anthracene, known for its robust C-H bonds, is a testament to its potential in mitigating toxic pollutants.
Real-World Applications and Impacts
The impact of this research extends beyond the laboratory; it opens avenues for industrial-grade metal catalysts that are capable of dismantling strong carbon-hydrogen bonds, which are often resistant to decomposition. This kind of technological breakthrough is vital as it aligns with global efforts to reduce pollution and embrace sustainable technological practices. Aromatic hydrocarbons, particularly those that are chemically stable and insoluble in water, represent a significant challenge in pollution cleanup. The newfound ability to decompose these substances effectively positions this catalyst as a game-changer in environmental remediation efforts.
A Vision for the Future of Chemistry
As emphasized by Professor Cho, the achievement of initiating reactions with a manganese(III) complex that includes two water molecules at lower temperatures represents an unprecedented milestone in catalyst design. This research not only contributes to our theoretical understanding of chemical processes but also sets a practical framework for future innovations in catalyst technologies. The focus on controlling manganese’s reduction potential serves as a beacon for aspiring researchers interested in crafting solutions for some of the most pressing environmental challenges of our time. In a world increasingly burdened by pollution, this catalyst exemplifies hope and influence, ushering in a new chapter in catalysis and environmental sustainability.

Leave a Reply