The accumulation of synthetic polymers in the environment has reached an alarming scale, with approximately 8.3 billion metric tons produced since the 1950s. Once hailed for their durability and versatility, these materials have been at the heart of a growing ecological crisis. A mere 600 million metric tons have been effectively recycled, leaving a staggering majority discarded or incinerated, generating significant environmental pollution. This stark reality underscores the urgency for innovative recycling solutions that can revitalize our approach to polymer waste management.
Innovative Research at the Forefront
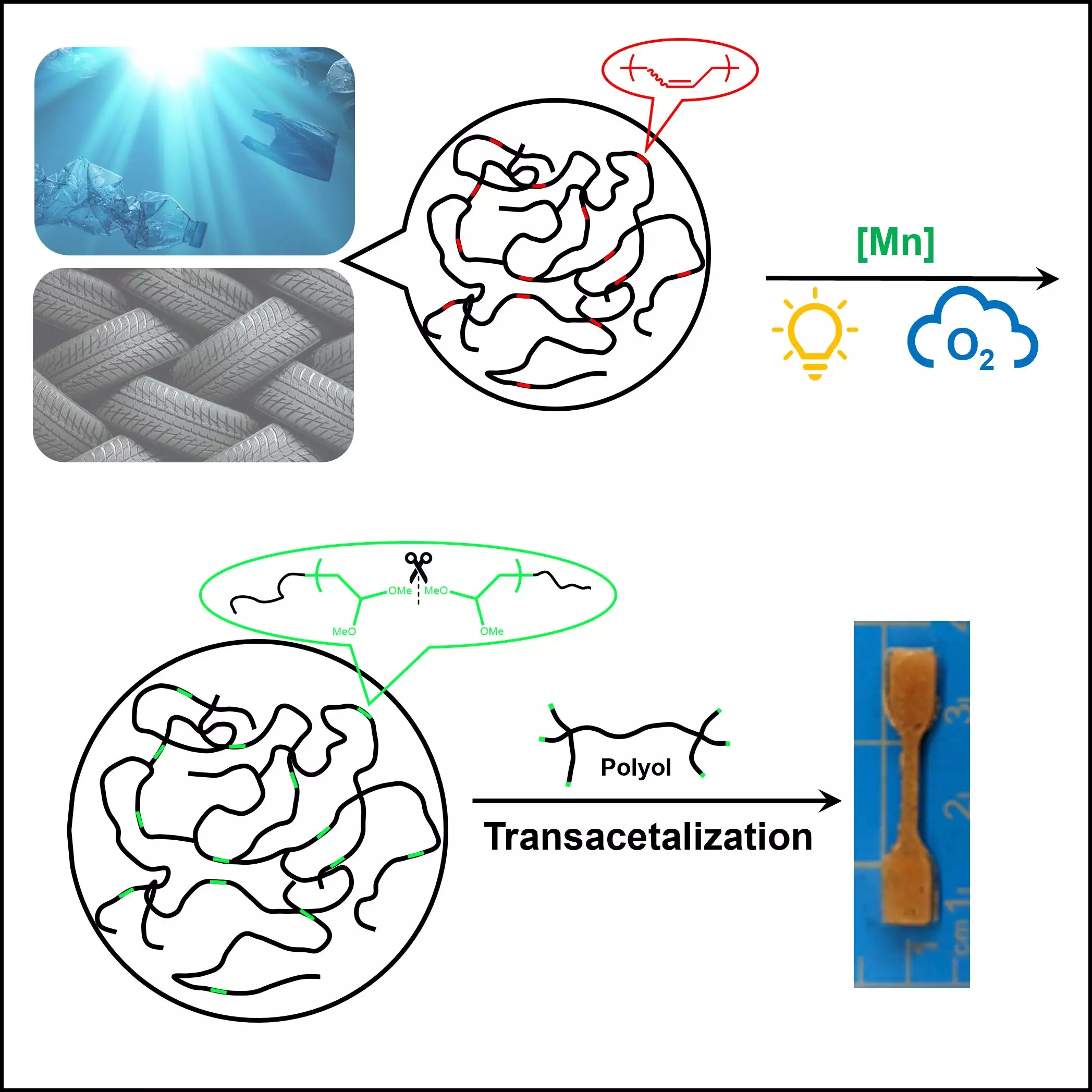
Dr. Junpeng Wang and his dedicated research team at The University of Akron have made a commendable stride toward addressing this pressing issue. Their recent publication in Cell Reports Physical Science unveils a groundbreaking technique for recycling unsaturated polymers, including rubber and commonly used plastics. This innovative method harnesses the combined power of oxygen and light, creating a controlled environment that enables the breakdown of polymers without resorting to the extreme conditions typically associated with traditional recycling processes.
Breaking Barriers in Polymer Recycling
Historically, recycling methods have been plagued by energy-intensive processes that not only produce hazardous by-products but are also impractical for large-scale implementation. Techniques like ozonolysis, while effective in certain contexts, fall short due to their high environmental cost and limited scalability. Wang’s research addresses these obstacles head-on, proposing a novel catalyst activated by light that operates at room temperature. This approach represents a significant paradigm shift, simplifying the recycling process and making it more sustainable.
Utilizing oxygen as the ideal oxidant for polymer degradation, Wang’s work offers an eco-friendly alternative to traditional methods. The abundance and accessibility of oxygen make it a compelling choice for sustainable practices in polymer recycling. By enhancing polymer reactivity through the introduction of unsaturation, the team has unlocked new pathways for effective degradation, fostering hope for managing polymer waste.
Implications for the Future
Wang and his team’s findings could serve as a catalyst for widespread advancements in recycling technology, potentially transforming not only the scalability of recycling efforts but also reshaping our understanding of polymer degradation mechanisms. The implications extend far beyond merely enhancing recycling rates; they pave the way for a future in which the lifecycle of synthetic materials is understood and managed more responsibly.
As the world grapples with the consequences of plastic pollution, the research from The University of Akron emerges as a beacon of hope. By setting a new standard for the management of unsaturated polymers, this study not only addresses a crucial environmental challenge but also inspires the development of further innovative strategies that harmonize human consumption with ecological sustainability. The potential to scale this method globally could signify a turning point in our ongoing battle against polymer waste, encouraging industries worldwide to rethink how they produce and dispose of plastics.

Leave a Reply