The quest to extract value from waste materials has always been a significant goal for chemists, particularly as the urgency to confront climate change intensifies. One groundbreaking development comes from an international consortium of scientists who are harnessing the power of electricity to innovate in the realm of chemical synthesis. Their recent research, published in the respected journal Nature Catalysis, showcases an exciting methodology for converting carbon dioxide—a notorious greenhouse gas—into methanol, a versatile liquid fuel. This work does more than merely add to existing knowledge; it opens the door to a future where waste could transform into energy, reinforcing the idea that innovation is the linchpin of our environmental response.
The key to this conversion lies in a particular molecular connection: cobalt phthalocyanine (CoPc). When optimized for efficiency and deployed on the unique architecture of carbon nanotubes—tiny cylindrical structures that boast remarkable electrical properties—CoPc demonstrates its potentiality. The integration of these materials with an electrolyte solution, under the influence of an electrical current, allows CoPc molecules to effectively harness electrons. This process enables the electrifying transformation of carbon dioxide into methanol and presents a tangible pathway to address one of the most pressing challenges of our time.
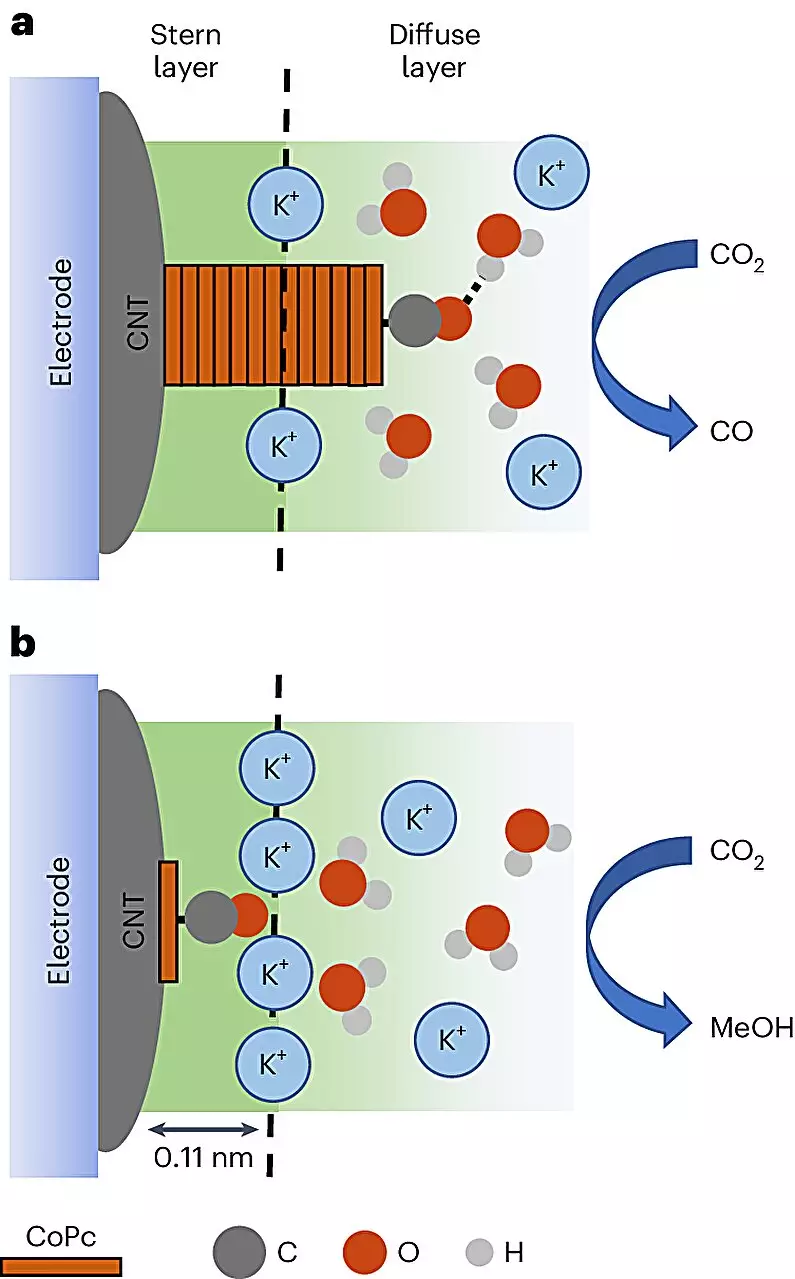
The Mechanics of Transformation
What sets this research apart is its innovative methodology for visualizing the reaction dynamics. By employing in-situ spectroscopy, scientists gleaned unprecedented insights into the conversion process, observing how CoPc molecules either shift to produce methanol or take a detour towards forming carbon monoxide, an unwanted byproduct. The revelation that reaction environment dictates the pathway taken is significant, illustrating that intricate adjustments to the CoPc distribution on carbon nanotube surfaces can enhance the likelihood of methanol production by a staggering eightfold.
This revelation is a game-changer. As Robert Baker, one of the study’s co-authors, eloquently articulated, the ability to convert carbon dioxide into various functional products is essential, and methanol stands out due to its high energy density. Its capacity to serve as an alternative fuel underscores a fundamental drive within the scientific community: the development of sustainable materials that mitigate environmental impacts while catering to energy demands.
From Empirical to Informed Optimization
Historically, the optimization of catalytic processes relied heavily on empirical methods. Experts would hone in on catalytic performance but remained in the dark regarding the underlying reasons for success or failure. Baker’s observations emphasize the need for comprehension beyond mere results. Thanks to advanced techniques—for which the research team state-of-the-art vibrational spectroscopy emerged as particularly crucial—their understanding of molecular interactions has deepened significantly.
Quansong Zhu, the lead author of the investigation, highlights another layer of complexity: the vibrational signatures observed indicate that identical molecules can react differently depending on their environmental conditions. This insight not only clarifies the mechanisms involved in methanol generation but also enhances the prospects for optimizing future chemical processes.
The Significance of Cations and Future Implications
Delving deeper into the intricacies of the reaction reveals additional findings that could propel this research into exciting new territories. The team noted that cation interactions served as supercharged facilitators for methanol formation, a discovery that prompts numerous questions regarding their roles in this type of chemical transformation. Moving forward, a better understanding of these cations could unveil groundbreaking applications, positioning methanol not just as a fuel alternative but as an important component in energy generation, heating, and possibly as a precursor for novel chemical products.
Methanol, derived from renewable electricity, is not merely a distant dream; it stands poised to revolutionize how we view energy sources for vehicles ranging from automobiles to ships, further decreasing our reliance on fossil fuels. As Baker suggests, the results of this research set a foundation for future explorations that could reshape our approach to energy solutions.
The Road Ahead: A Catalyst for Change
As these researchers continue to unravel the complexities of chemical reactions on a molecular level, the future of energy production appears increasingly bright. This study not only propels our understanding of carbon dioxide transformation but also instills hope for sustainable practices that future generations can rely on. With ongoing investigations, the quest for innovative applications of these findings is just beginning, promising a world that is not only more efficient but also planet-friendly, paving the way for a cleaner, greener planet.

Leave a Reply