Scientific research in molecular chemistry continuously ventures into uncharted territory, unveiling possibilities that might radically transform various fields. A recent study led by Professor Ingo Krossing at the University of Freiburg’s Institute of Inorganic and Analytical Chemistry has yielded promising results in the realm of oxidation potentials, particularly regarding positive ions. This breakthrough not only showcases the innovative spirit of modern chemistry but also sheds light on the strategies required to overcome longstanding challenges in redox reactions.
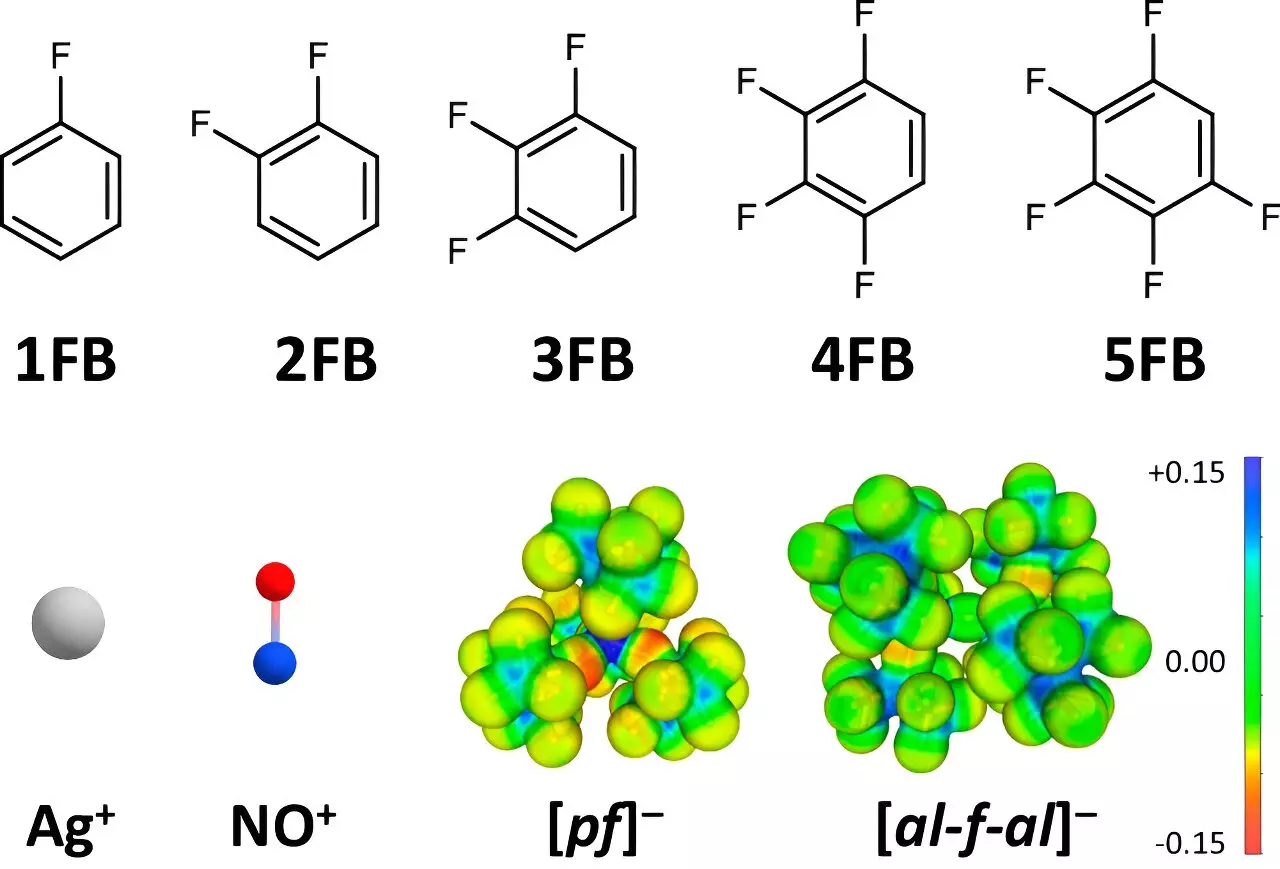
Traditionally, the oxidation potential of positive ions like Ag+ and NO+ has been a limiting factor in their application within chemistry and materials science. These cations, due to their small size and considerable charge density, typically exhibit oxidation potentials that hover around +0.65 to +1.0 V compared to the ferrocene/ferrocenium (Fc+/0) reference. However, the collaborative efforts in this research group have resulted in a remarkable leap forward, demonstrating potentials soaring up to +1.50 and +1.52 V with the application of specially designed solvent systems.
This advancement was possible thanks to a meticulous selection of weakly coordinating anions and solvents. By analyzing their interactions, the researchers have managed to enhance the oxidation power of these ions significantly, allowing for the execution of previously unattainable redox reactions.
A pivotal component of this study was the strategic employment of fluorinated benzene derivatives as solvents. The choice of these specific organic compounds is not arbitrary; it emerges from a deeper understanding of their dielectric properties as established in collaboration with Dr. Johannes Hunger from the Max Planck Institute for Polymer Research. The research indicates that certain multi-fluorinated aromatic solvents demonstrate significantly higher dielectric constants compared to traditional solvents like dichloromethane or acetone.
Such differences in solvent characteristics have profound implications. When these fluorinated derivatives act as solvents, the interaction with the positively charged ions evolves, becoming less pronounced as the fluorination increases. This allows the positive ions to remain in a less disturbed state, thus enhancing their oxidation capabilities.
The implications of these findings extend far beyond merely improving existing redox reaction methodologies. With a greater potential for oxidation, this research paves the path toward novel applications in electrocatalysis, a field that is becoming increasingly crucial in energy conversion and storage technologies. The ability to facilitate reactions that were once deemed too difficult to achieve could lead to advancements in energy storage systems or the development of more efficient chemical processes.
Moreover, the researchers anticipate that this work could revolutionize the design of redox mediators and shuttles, which play a critical role in various industrial chemical processes. The enhanced oxidation capabilities of positively charged ions could introduce new pathways for chemical reactions, thus accelerating the rate and efficiency of various syntheses.
This significant boost in understanding and manipulating oxidation potentials is a testament to collaborative scientific endeavors. The integration of knowledge from both the University of Freiburg and the Max Planck Institute illustrates the essential nature of interdisciplinary collaboration. Each expert brought different strengths to the table that culminated in a groundbreaking study published in Nature Communications.
By strategically employing weakly coordinating anions and innovative solvent systems, Professor Krossing and his team have marked a significant milestone in the field of molecular and coordination chemistry. Not only does this research advance our understanding of oxidation potentials, but it also opens new avenues for exploration in chemical synthesis and electrocatalysis, potentially reshaping the landscape of materials science for years to come.

Leave a Reply