In the ever-evolving realm of chemistry, the quest for energy-efficient synthesis methods is paramount. New findings from researchers at RIKEN herald a significant advancement in this goal, showcasing an innovative method for utilizing dinitrogen (N₂), the most abundant molecule in our atmosphere, in the synthesis of valuable compounds. This article delves into the implications of their discovery and the potential it holds for both industry and sustainability.
The Dilemma of Dinitrogen Utilization
Dinitrogen is a resource that is nearly omnipresent, constituting nearly 80% of the Earth’s atmosphere. Despite its abundance, harnessing its potential in chemical reactions has long been hampered by the stability of its triple bond, which renders direct reactivity difficult. Traditional methods, such as the Haber-Bosch process, require breaking this bond to synthesize ammonia. This intermediate step, while integral for the production of alkyl amines and polymers, introduces inefficiencies and significant energy costs to the chemical synthesis landscape.
The challenges extend beyond the mere reactivity of dinitrogen; they also encompass the necessary pre-activation of alkene components for successful synthesis. Conventional approaches typically transform alkenes into alcohols or carboxylic acids, resulting in cumbersome processes that demand additional time and energy resources. As articulated by Takanori Shima of RIKEN’s Center for Sustainable Resource Science, there is an urgent need for more straightforward methodologies that can utilize dinitrogen and alkenes under mild conditions without extensive pre-treatment.
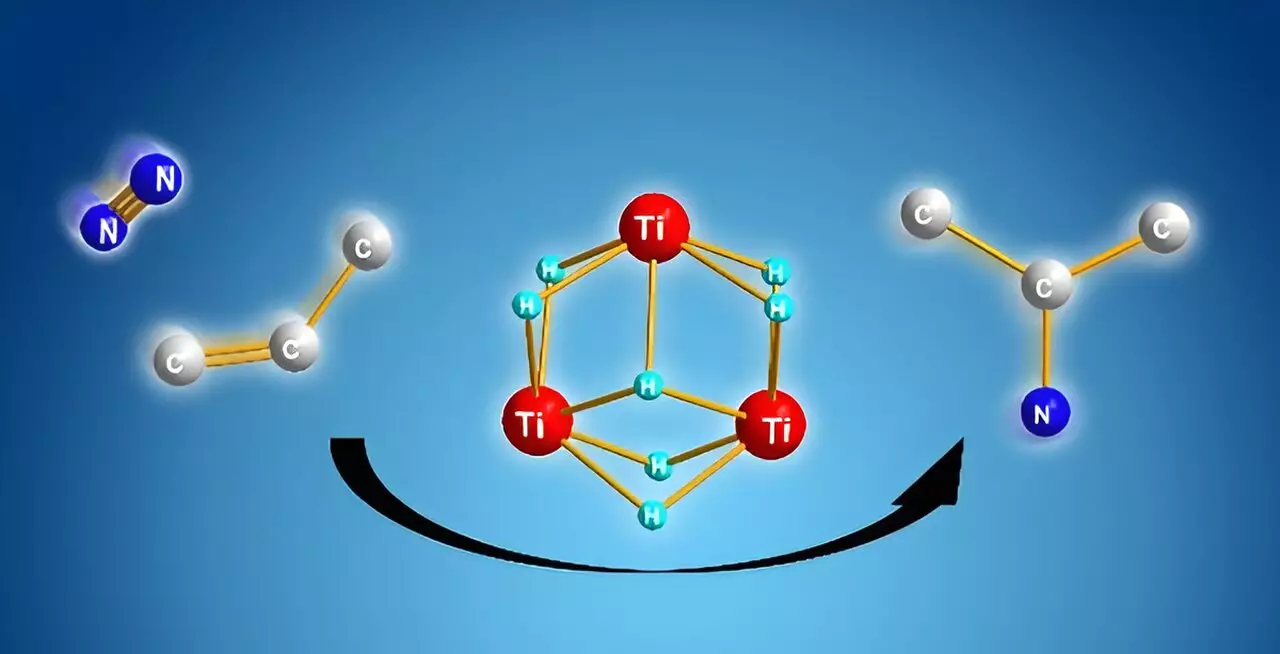
The study led by Shima and his colleagues introduced an innovative approach using titanium polyhydrides—complexes wherein titanium atoms are interconnected by hydrogen atoms. This unique structure demonstrates a remarkable reactivity towards stable molecules, including dinitrogen and benzene. What sets titanium polyhydrides apart is their ability to facilitate reactions that were previously considered highly challenging.
Through extensive research, the team discovered that these complexes could activate alkenes effectively, while retaining several titanium-hydride units post-reaction. By introducing dinitrogen to these activated alkenes, the remaining titanium-hydride units work collaboratively to cleave the dinitrogen triple bond, subsequently forming meaningful nitrogen-carbon bonds. This novel mechanism results in the efficient synthesis of alkyl amines, a chemical class valuable for pharmaceuticals and industrial applications alike.
A deeper dive into the underlying mechanics reveals a fascinating aspect of the reaction process. Computational analysis suggests that once both substrates—alkenes and dinitrogen—are activated, the formation of nitrogen-carbon bonds is favored over other potential pathways like nitrogen-hydrogen or carbon-hydrogen bond formations. This preference indicates that the titanium polyhydride framework effectively guides the reaction energetically, favoring the pathways that yield the desired alkyl amines.
Such insights are not only scientifically intriguing; they present a broader canvas for catalytic synthesis methodologies that could redefine how chemical reactions are conducted in the future. The demonstration that these transformations can occur within the titanium polyhydride framework under mild conditions opens new avenues for research, highlighting the potential for scalable applications in industrial settings.
Shima and his team are now setting their sights on exploring the potential of this transformative process as a catalytic reaction. The prospect of developing a catalytic system using titanium polyhydrides not only promises to enhance efficiency and sustainability in chemical processes but also aligns with global efforts toward greener industrial practices.
As chemists continue to innovate in leveraging abundant atmospheric resources, the implications of this research could reverberate through multiple sectors, facilitating the creation of drugs, polymers, and numerous other compounds with reduced energy inputs. The intricate dance between dinitrogen and titanium polyhydrides signifies a promising leap towards achieving a more sustainable and efficient future in chemical synthesis.
The research from RIKEN illuminates the potential of using dinitrogen in innovative ways while addressing energy consumption in chemical processes. The exploration of titanium polyhydrides could pave the way for a new chapter in synthetic chemistry—one that emphasizes efficiency, accessibility, and sustainability.

Leave a Reply