Fuel cells represent a pivotal technology in the quest for sustainable energy solutions. By converting chemical energy directly into electricity through electrochemical reactions, these cells have the potential to power a myriad of applications ranging from electric vehicles to portable electronics. The allure of fuel cells lies in their environmental benefits, chiefly that they produce electricity without combustion, thus minimizing air pollutant emissions. However, despite their potential, the widespread adoption of fuel cells has been stunted due to various limitations, primarily the dependence on expensive materials and precious metal catalysts that contribute to high production costs.
The need for more affordable and effective alternative solutions has led researchers worldwide to explore anion-exchange-membrane fuel cells (AEMFCs). These fuel cells are distinguished by their reliance on Earth-abundant materials, which could facilitate broader usage in diverse energy applications. Nonetheless, the field has encountered significant challenges, including the tendency of non-precious metal catalysts to experience self-oxidation, leading to irreversible degradation of the cells. Thus, the search for reliable catalytic materials remains a critical issue.
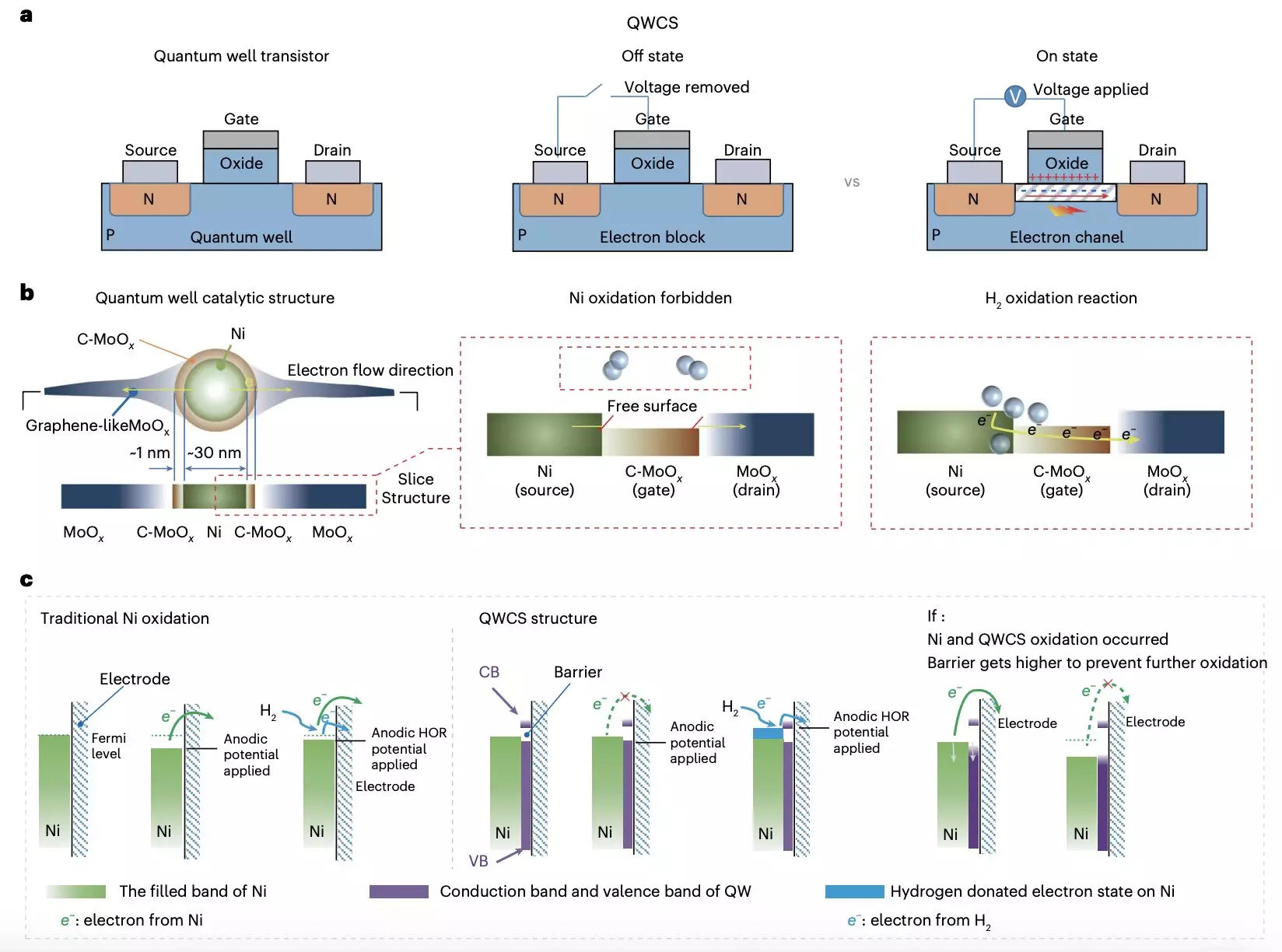
Recent advancements from research teams at Chongqing University and Loughborough University offer a promising pathway to address some of the deficiencies associated with conventional AEMFC technology. Their innovative approach centers around a unique catalytic structure known as the quantum well-like catalytic structure (QWCS), which incorporates quantum-constrained nickel nanoparticles. This design not only aims to enhance catalytic efficiency but also seeks to mitigate the oxidation problems that have historically hindered the stability of non-precious metal catalysts.
According to the researchers, traditional non-precious catalyst materials often succumb to self-oxidation, resulting in stark reductions in performance. Their work demonstrates that by employing the QWCS, nickel nanoparticles can be atomically confined within a carbon-doped molybdenum oxide (C-MoOx) heterojunction. This structure not only optimizes electron transfer rates during hydrogen oxidation reactions but also protects the nickel catalyst from degradation.
What sets the newly designed Ni@C-MoOx catalyst apart is its ability to selectively facilitate the transfer of electrons derived from the hydrogen oxidation reaction while simultaneously preventing the transfer of electrons from the nickel catalyst itself into the QWCS’s low-energy valley. This innovation significantly enhances the stability of the catalyst against detrimental electro-oxidation processes, thereby extending the operating life of AEMFCs.
The team’s experimental results signify a breakthrough in the use of QWCS for improving the longevity and performance of fuel cells: Their Ni@C-MoOx catalyst maintained remarkable hydrogen oxidation reaction (HOR) stability after 100 hours of continuous operation under rigorous testing conditions. Additionally, the developed AEMFC demonstrated a superior specific power density of 486 mW mgNi-1, illustrating not only its efficiency but also its resilience during shutdown-start cycles—a critical factor often associated with the failure of conventional systems.
The Road Ahead: Implications for Future Energy Technologies
The implications of these findings reach far beyond the laboratory. By enabling the use of cost-effective, non-precious metal catalysts that exhibit enhanced stability, the research signals a transformative potential for AEMFC technology. The Ni@C-MoOx catalyst could become a cornerstone in the development of next-generation fuel cells, substantially lowering production costs while maintaining robust performance.
Moreover, the strategic design principles unveiled by this research may extend their relevance to various other applications within the energy sector, potentially informing the development of new catalysts that leverage quantum confinement effects to counteract the issues commonly associated with non-precious materials. As energy demands continue to rise globally, such advancements will be crucial in paving the way toward more sustainable and efficient energy solutions.
The synthesis of advanced catalysts such as those developed in this research is not merely an incremental improvement; it marks a significant step forward in our understanding and utilization of fuel cell technology. By addressing the key challenges of cost and stability, researchers are setting the stage for broader adoption of fuel cells in the green energy landscape.

Leave a Reply