Nitrate contamination in drinking water has become a pressing concern that poses severe risks not only to environmental stability but also to human health. The urgency of this issue requires innovative solutions capable of effectively mitigating the problem. Traditional methods, while helpful, often fall short in both efficiency and effectiveness, leading researchers to explore new techniques. Yale researchers, led by Lea Winter, assistant professor of chemical and environmental engineering, have made significant strides in this field through the development of an electrified membrane system designed for rapid nitrate elimination. This breakthrough offers promise in the quest for clean and safe drinking water.
The Limitations of Traditional Methods
Historically, the approaches to removing nitrates from water have focused on two main strategies: separation and destruction. Separation can create a concentrated waste stream, presenting further risks of recontamination in the environment. On the other hand, traditional destruction methods, primarily biological denitrification used in wastewater treatment plants, savor their own set of challenges. These processes are notoriously sensitive to variations in conditions such as pH, temperature, and microbial health. Even the slightest deviation can wreak havoc on the ability to cleanse contaminated water effectively. The slow nature of these biotechnological methods further exacerbates the issue, highlighting the need for an industry shift towards more reliable technologies.
Electrocatalytic Processes: The Faster Alternative
To address these shortcomings, scientists have shifted their focus towards electrocatalytic processes as a potential remedy. Unlike traditional methods, these newer techniques capitalize on electrochemical reactions, allowing for faster nitrate reduction. However, the classical electrochemical designs utilize flat plate electrodes, which have been shown to struggle with efficient nitrate transport to the electrode surface. The challenge lies in that the closest layer of fluid to these electrodes, known as the “boundary layer,” can hinder rapid reaction rates due to its considerable thickness—on the order of 100 micrometers. This barrier leads to diminished effectiveness in nitrate conversion, renewing the quest for a more effective mechanism.
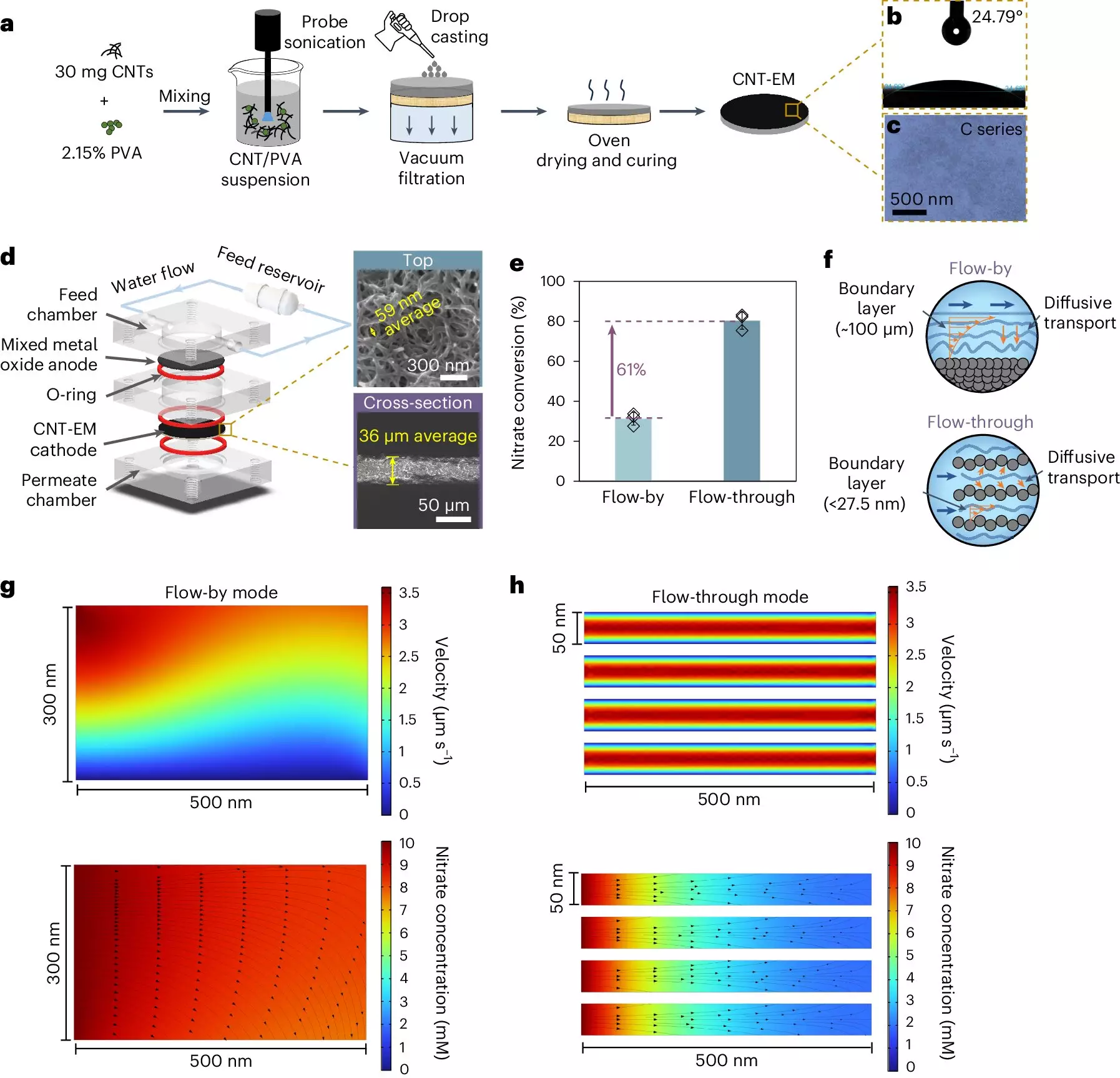
Innovative Electrified Membranes
In response to these challenges, Winter’s lab has developed electrified membranes that significantly enhance nitrate removal efficiency. These membranes utilize carbon nanotubes embedded within a polymer matrix, providing a platform that is essentially a game changer in the nitrate removal landscape. The key innovation rests in the remarkably small pore sizes of these membranes—around 50 nanometers, in stark contrast to the typical dimensions found in conventional systems. This drastically reduces the sluggish boundary layer effect, thereby promoting quicker nitrate transport to the reactive sites on the membrane. The implications of this advancement are profound; they not only bolster the reaction rate but also sidestep the common pitfalls associated with larger-pore systems.
A Breakthrough in Speed and Efficiency
The performance of Winter’s membranes is nothing short of astounding. In traditional electrochemical systems, achieving an 80% or 90% nitrate removal rate could take several hours. However, under the new methodology, similar results can be accomplished in as fast as 15 seconds. This drastic reduction in processing time marks a considerable advancement in water treatment capabilities, enabling quicker responsiveness to nitrate contamination crises. The ability to handle lower concentrations of nitrate—which are often more representative of real-world conditions—further underscores its practical applicability.
Real-World Testing and Future Implications
In a bid to validate their innovative technique, Winter and her research team conducted practical tests with water samples from Lake Wintergreen, deliberately adding nitrates to assess the efficacy of their membranes. The initial results reveal promising potential, showcasing that even at typically low concentrations, the technology holds water—literally and metaphorically. As nitrate contamination increasingly threatens ecosystems and human health, this breakthrough could facilitate a major enhancement in drinking water safety.
Winter’s pioneering work in carbon nanotube-based membranes not only demonstrates the power of engineering innovation but also signifies a beacon of hope for communities facing the repercussions of toxic water supplies. As this young scientist and her team continue to explore the intricacies of this technology, the future of water treatment looks increasingly bright.

Leave a Reply