In an impressive stride towards advancing organic chemistry, Professor Max Martin Hansmann and his dedicated team at the Department of Chemistry and Chemical Biology have unveiled an innovative reagent designed for the selective addition of carbon atoms to various molecular frameworks. This groundbreaking research, which was reported in esteemed journal Science, not only highlights Hansmann’s commitment to his field but also underscores the significance of strategic funding, as this endeavor was part of his ERC Starting Grant awarded in 2022—an opportunity that has allowed him to catalyze new discoveries.
Leading the charge in this academic expedition is Dr. Taichi Koike, a postdoctoral researcher whose work as the first author of the study reflects a height of creativity and scientific prowess within the realm of molecular science. His role as an Alexander von Humboldt Research Fellow at Hansmann’s laboratory further showcases the collaborative efforts that fuel progress in complex organic synthesis.
The Elegance of Single-Atom Modifications
The elegance of Professor Hansmann’s work rests on the premise of precise modifications at a single-atom level. The ability to manipulate molecular structures with such precision is considered one of the most sophisticated techniques in organic chemistry, leading to an enticing prospect: the synthesis of complex pharmaceuticals through shortened and optimized sequences. Such advancements promise not just to streamline research processes, but also to pave the way for the development of novel compounds with potentially transformative medical benefits.
However, the journey towards achieving this precision is fraught with challenges, particularly in developing reagents that can effectively introduce carbon atoms with selectivity. Hansmann’s success in synthesizing a new type of reagent positions this research as a key to unlocking the complexity of molecular design, allowing chemists to explore previously uncharted territories in synthetic methodology.
Innovative Synthetic Strategies
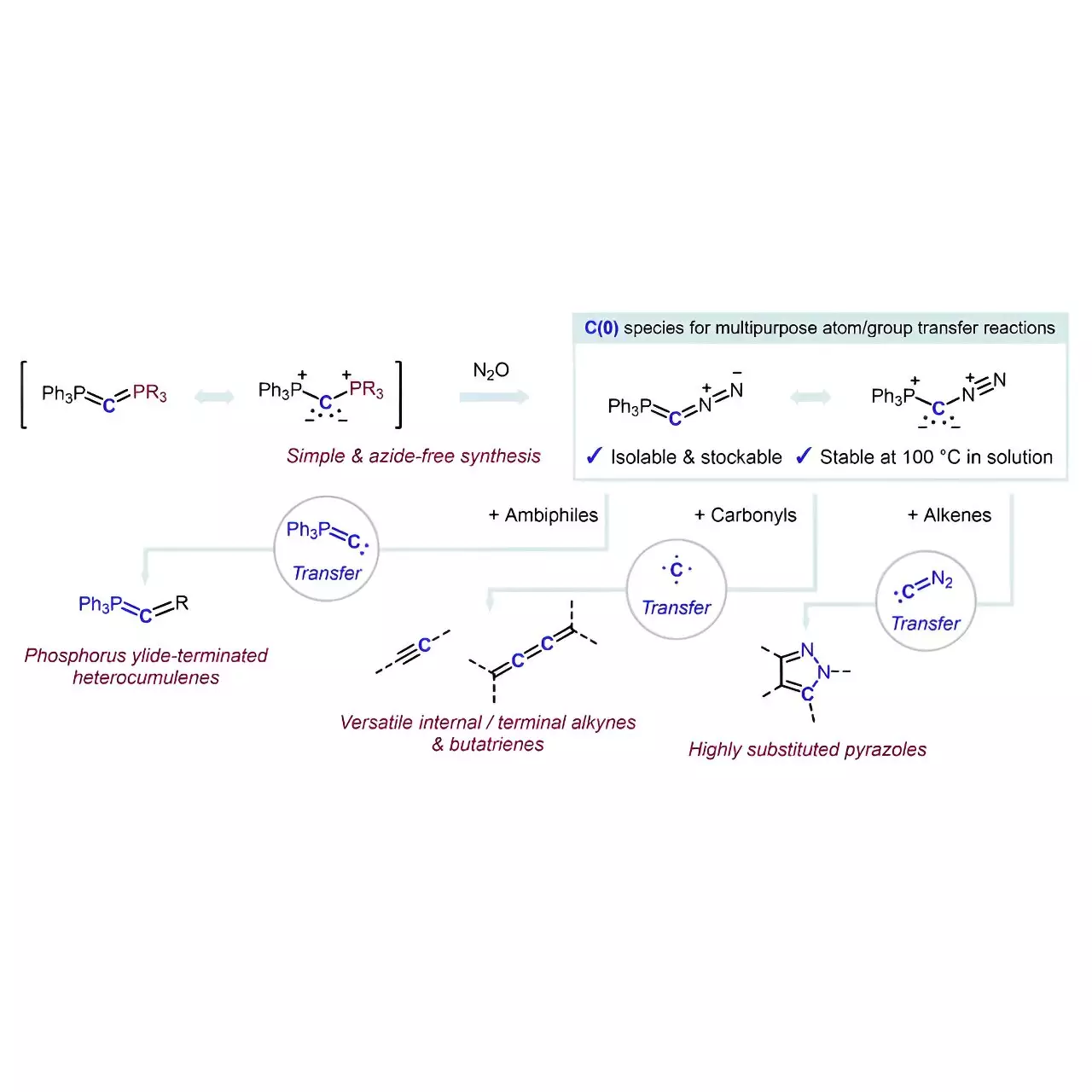
What sets this achievement apart is the innovative approach taken by Hansmann and his team to stabilize a carbon atom through coordination with two neutral electron-donating groups. This clever stabilization has given rise to a species known as carbones (L1→C←L2), a class that had received limited attention in the past as a viable carbon source. By overcoming the challenges inherent to using carbones—often fraught with stability concerns and practical limitations—the researchers have developed a reagent where a carbon atom is encased between two labile groups: PPh3 and N2.
The synthesis of the crystalline and isolable reagent Ph3P=C=N2 is a notable highlight of the research. It emerged from a simple yet efficient formal exchange reaction involving carbodiphosphorane (Ph3P=C=PPh3) and nitrous oxide (N2O), aptly sidestepping the complications posed by azides commonly associated with diazo compounds—issues that often raise safety flags in laboratory settings. Such methodological elegance not only promotes safety but also amplifies the accessibility of this reagent for broader adoption across organic chemistry research.
Pioneering New Pathways in Molecular Synthesis
The practical applications of Hansmann’s new transfer reagent are expansive. The effectiveness of Ph3P=C=N2 as a selective transfer reagent allows fragments from the carbon atom to transfer without additional reagents, demonstrating commendable efficiency in reactions. For example, when transferring to ambiphiles, the process produces phosphorus ylide-terminated heterocumulenes, while interactions with alkenes yield multi-substituted pyrazoles. Most intriguingly, its reaction with carbonyl compounds facilitates direct carbon atom transfer, resulting in a variety of alkynes or even butatrienes—molecular structures that can hold immense synthetic and therapeutic potential.
What stands out in Hansmann’s research is not only its immediate implications for organic synthesis but also its ability to inspire future inquiries into the mechanics of carbon atom transfer. The foundation laid by this study is likely to drive new exploration into the creation and functionalization of complex molecular structures, opening avenues that were previously limited due to the challenges associated with carbon atom incorporation.
In a landscape increasingly rich with scientific breakthroughs, Hansmann and Koike’s work is poised to transform the paradigms of organic chemistry—one carbon atom at a time. The future of pharmaceutical development and synthetic chemistry seems brighter with each advance achieved in precision with this remarkable new reagent.

Leave a Reply